Replacing fossil fuels with electricity or biofuels does not mean reliance on gas and oil will go away overnight. In addition to transportation fuels, petroleum is refined into hundreds of petrochemicals used in everything from plastics and pharmaceuticals to carpets and fertilizers, and currently there are no easy ways to produce most of these chemicals without using oil.
But Jay and Cynthia Ihlenfeld Professor Brian Pfleger and his colleagues in the Department of Chemical and Biological Engineering recently developed a pathway with the potential to convert biomass, like wood chips or grasses, into a widely used industrial solvent and aim to expand this work to produce an array of sustainable chemicals.
 Brian Pfleger
Brian Pfleger
The project was the brainchild of Pfleger and Ernest Micek Distinguished Chair in Chemical and Biological Engineering James Dumesic. “Jim does a lot of the same types of things I do, but from a metal-based or chemical-based catalysis perspective, whereas I’m using living organisms or bio-catalysts,” he says.
In particular, Dumesic’s group has developed chemical processes that convert lignocellulosic biomass, essentially dried plant matter, into levulinic acid, a five-carbon organic acid with a high potential to become a platform precursor to biofuels and other chemical products.
By using microorganisms to further process the acid into usable chemicals, Pfleger believed he could keep the process sustainable from start to finish. “What we were hoping to have then was a route to go from a green material that was biomass-derived to a commodity chemical that would have otherwise been produced from petrochemical or other traditional refineries,” he says. “But first we needed to figure out what organism can process those chemicals and how they do that.”
Luckily, researchers had previously identified an organism that could “eat” levulinic acid, a bacteria called Pseudomonas putida KT2440 that uses the acid to produce more cells. Pfleger and members of his lab were interested in identifying the specific genes that enabled this ability.
Pfleger describes the effort to find the genes, led by Jacqueline Rand (PhDChE ’17) and published in Nature Microbiology, “a tour de force of classic and modern gene discovery techniques.”
A team of undergrad researchers screened a 6,000-sample transposon library, looking at how transposons, or “jumping” genes that turn various genetic segments on or off, affected the organism’s ability to process levulinic acid. If a gene was turned off and the bacteria could no longer “eat” the acid, they knew it was likely involved in metabolizing the compound.
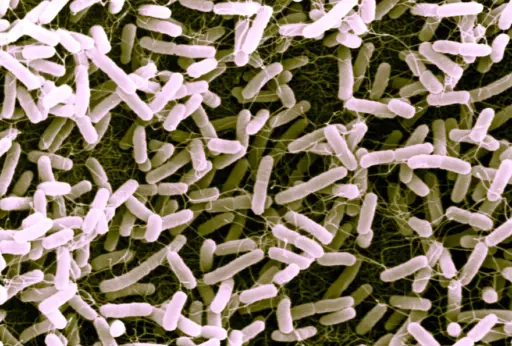 The researchers were able to modify E. coli (pictured) so that it converted levulinic acid into methyl ethyl ketone, otherwise known as butanone, a common industrial petrochemical solvent.
The researchers were able to modify E. coli (pictured) so that it converted levulinic acid into methyl ethyl ketone, otherwise known as butanone, a common industrial petrochemical solvent.
“We zeroed in on a couple of hits from that library and found this series of co-regulated genes that are turned on and off together in the presence of levulinic acid,” Pfleger says. “And so, we were able to go through with a classic set of studies where we knocked out each of these individual genes and saw what the organism could or could not do.”
Working with Daniel Amador-Noguez in the Department of Bacteriology, the team used mass spectrometry and other techniques to track what, exactly, the enzymes were doing to process levulinic acid. “From that we were able to sleuth out this whole metabolic pathway,” Pfleger says. “It’s a series of reactions that take the levulinic acid, the molecule that we were going to start with from Jim Dumesic’s group, and get it to these two molecules called acetyl-CoA and propionyl-CoA that are in the core of metabolism. And so, cells can take these molecules wherever they want.”
The next step was to take those genes and place them into E. coli, a better understood model organism, to see if they could engineer it to handle levulinic acid as well. “Unfortunately, it didn’t just work right off the bat, as is so often true,” Pfleger says. “There were enzymes that were already in E. coli’s repertoire that were conflicting with the activity of these others.”
To solve the problem, the team conducted an evolutionary study, slowly increasing the concentration of levulinic acid the E. coli received while decreasing other inputs until they evolved and isolated a strain that was able to grow on the levulinic acid alone.
Collaborating with colleague Jennifer Reed, the Harvey D. Spangler Professor in chemical and biological engineering, Pfleger’s team was able to put all of the enzymes involved in the metabolism in a genome scale model, which allowed them to search for combinations of enzymes that force the metabolism in desired directions. With that information, they were able to modify the E. coli so that it converted the levulinic acid into methyl ethyl ketone, otherwise known as butanone, a common industrial petrochemical solvent.
Butanone is not the only chemical that can be made by this process. “This is the power of metabolic engineering,” Pfleger says. “Once those feedstocks are in the cell’s metabolism, they can be directed, like we did to methyl ethyl ketone, to many, many other compounds.”
And that, he hopes, will be a sustainable paradigm shift when it comes to the production of commodity chemicals. “In a petrochemical refinery, you have a list of compounds that are in the barrel of oil and you do some reactions and you make some chemical conversions. But basically, what you’re trying to do is find a use for every single one of those molecules,” he says. “Here, we’re starting with sugar, or in this case levulinic acid, and we can go to virtually any molecule a cell could make.”